Menu

Established in 2004, CIDP (Centre International de Développement Pharmaceutique) is an international Contract Research Organisation (CRO) that carries out high R&D activities for pharmaceutical, medical device, nutraceutical and cosmetic industries.

USE TEST
The tests involve the assessment of the tolerance of the cosmetic products under the supervision of medical experts for a defined duration of time. In addition, various subjective parameters such as stinging, burning and skin tightness can be evaluated using standard assessment criteria. The tests can be tailor made for a duration of 14, 21 or 28 days.
Under Dermatological Control
- Test design for claims such as tested under dermatological control
- Test panel include sensitive skin, reactive skin, atopic and atopic history, etc…
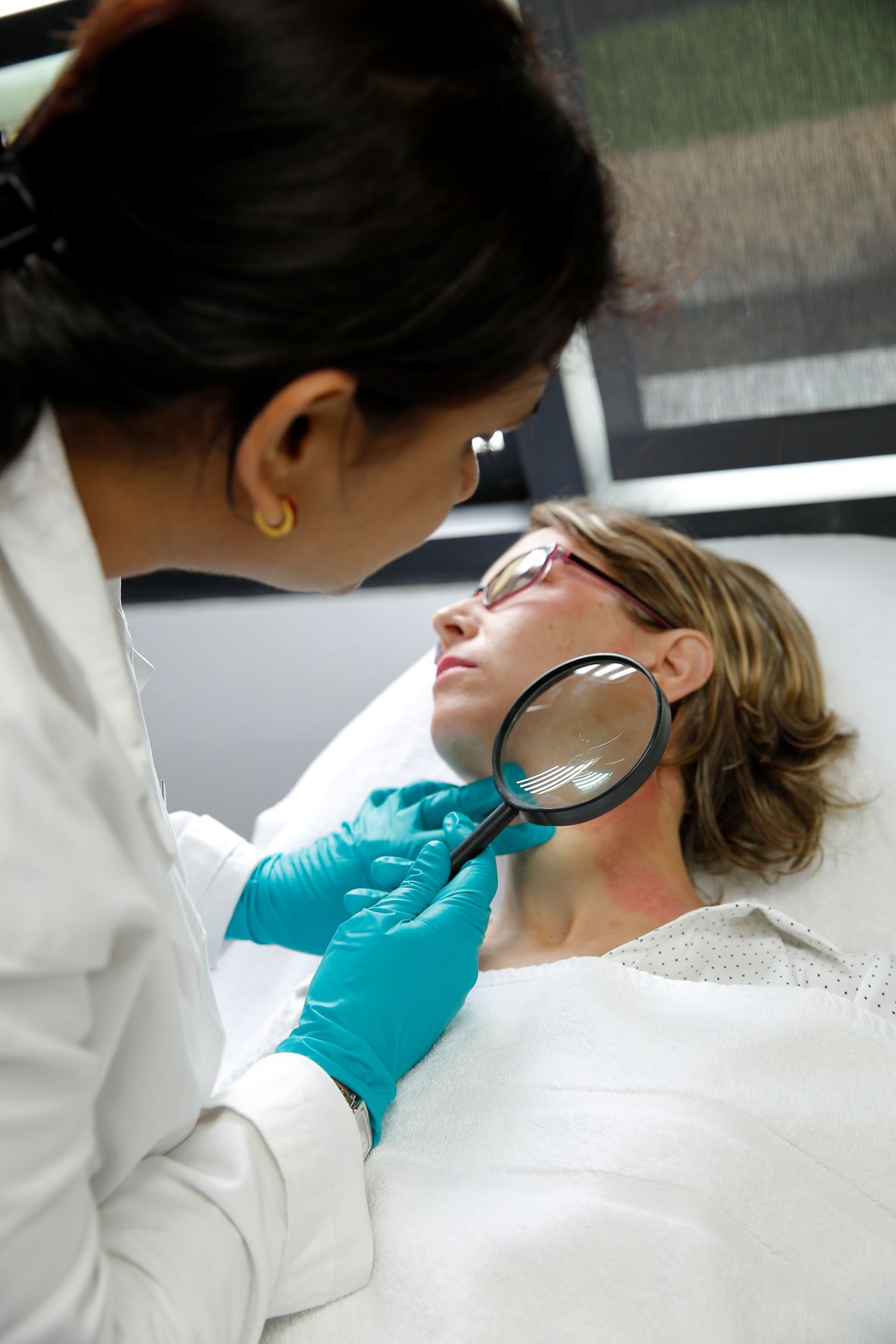

Under Ophthalmological Control
- For claims such as tested under ophthalmological control, “No Tears” or “does not sting eyes”
- Test panel include sensitive eyes, lens wearers, etc…
- Various testing methodologies such as:
- Stinging test
- Human Ocular eye instillation
Under Paediatrician Control
- For claims such as tested under paediatrician control, safe for use in children
- Test panel includes new-born, baby and child subjects
